1. BREATHE EASY

What are some forms of air pollution?

Our atmosphere is vital to all living things on our planet. How much do you know about the air we breathe?
Discover-AQ campaign to use satellites to monitor air quality

What are some forms of air pollution?

For how long do dust and soot generally remain in the air?

What type of pollution is not pumped out directly from cars and trucks?


Which type of air pollution is most difficult to measure from space?


Is ozone good or bad for humans?

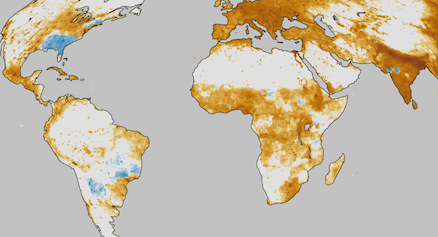
How far can air pollution travel from its source?


Is air quality in the U.S. better or worse on average than it was 20 years ago?


In the 10,000 years before the Industrial Revolution in 1751, carbon dioxide levels in the air rose by less than 10 percent. Since then they’ve risen by:

Which instrument, launching in 2022, will trace the origin and composition of mineral dust that can affect climate, ecosystems, air quality, and human health?

What are the strongest sources of nitrogen dioxide today?
