6 min read
Sizing Up Humanity's Impacts on Earth's Changing Atmosphere: A Five-Part Series
As discussed earlier in this feature series (see Parts One and Two), Earth’s atmosphere is largely able to cleanse itself of pollutants, but there are a few things that humans have produced that are much more long-lived when emitted into the atmosphere, degrading its quality and creating harmful environmental effects.
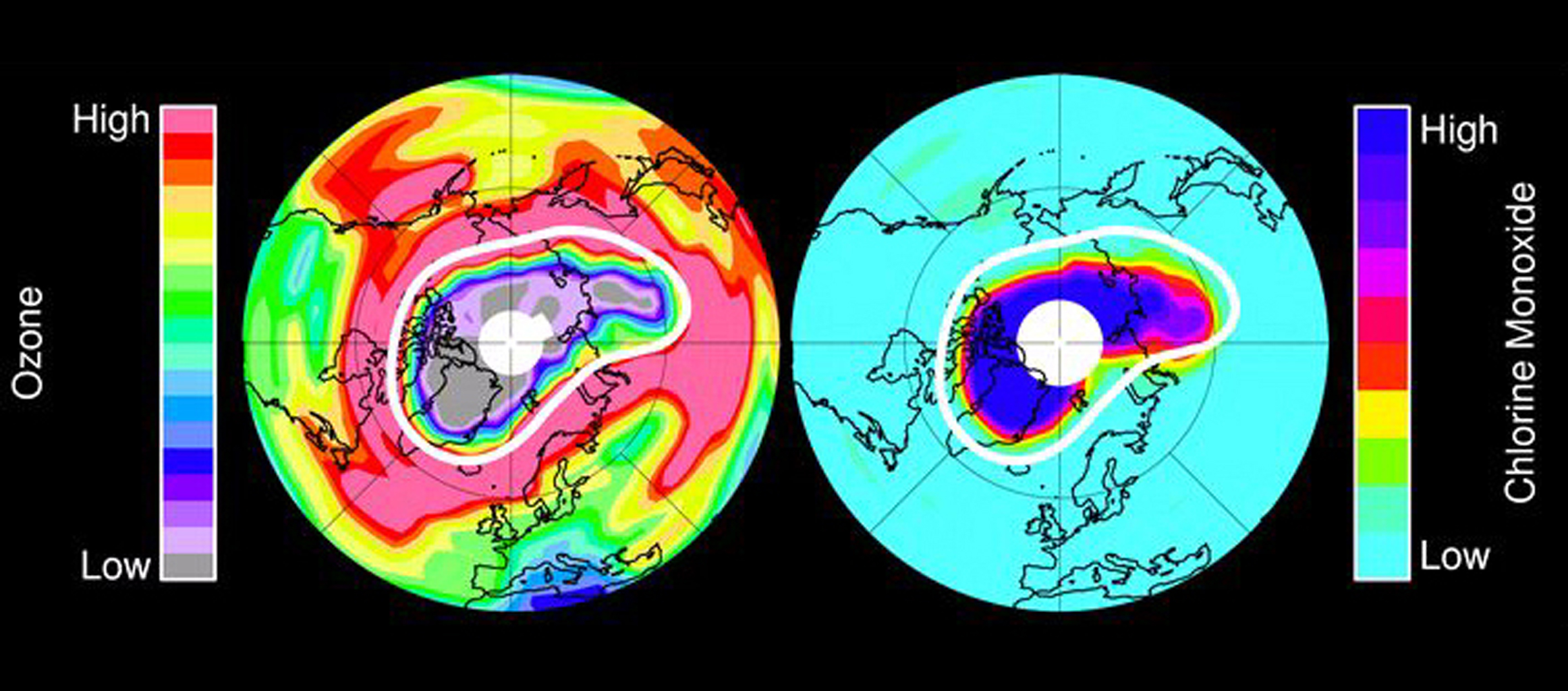
One such family of chemical compounds is chlorofluorocarbons (CFCs), whose contribution to depleting ozone in Earth’s upper atmosphere has led to large springtime decreases in ozone around Earth’s polar regions, especially over Antarctica, a phenomenon known as the ozone hole that was first reported in 1985. But, as NASA atmospheric scientist Nathaniel Livesey explains, today, thanks to the phase-out of CFCs, Earth’s ozone hole is in recovery. He says the turnaround provides a great example of what humans can do when they work together to solve a global atmospheric problem.
“Humans produced a lot of CFCs from the 1950s through the early 1990s that were useful for a variety of purposes and widely adopted around the world,” said Livesey, principal investigator for the Microwave Limb Sounder (MLS) instrument on NASA’s Aura satellite at NASA’s Jet Propulsion Laboratory in Pasadena, California. The CFCs were added to the atmosphere at the parts per billion level. “But CFCs were also very effective at depleting stratospheric ozone, which protects us from harmful solar ultraviolet radiation, and their use created a hole in Earth’s stratospheric ozone layer. Luckily, we were able to identify the problem in time and come to a worldwide agreement, the Montreal Protocol, which phased out their use.”
NASA
Under the Montreal Protocol, which was finalized in 1987, and its 2016 amendment, a multi-phased plan was implemented involving the use of hydrochlorofluorocarbons (HCFCs), which aren’t as damaging to the environment as CFCs and could be used in the same equipment, since their chemical structure is very similar to CFCs. But HCFCs also contribute to ozone layer destruction, albeit at a smaller rate than CFCs did, as well as to global warming, so their use is also being gradually phased out over the next decade.
While the Montreal Protocol is a great success story, Livesey cautions that tackling Earth’s carbon dioxide and methane emission problems will be more difficult to address.
“People everywhere used CFCs but, in actuality, there were only about four companies in the world that actually produced them,” he said. “With carbon dioxide, the problem is much more complex. All of us produce carbon dioxide. And there are way more coal-burning power plants than there ever were CFC plants. Methane emissions resulting from human activities are also a major contributor. So it’s very hard to point to one thing to fix the problem like we could with CFCs.”
Livesey says Earth’s recent temperature increases simply cannot be explained without accounting for human emissions of carbon dioxide, which builds up over time and has a long life once emitted into the atmosphere. To those who claim that carbon dioxide, methane and other greenhouse gases don’t have a significant impact on global warming, he offers a simple scientific experiment.
“Take a gallon of water and put a drop of food coloring in it. You’re going to immediately notice a change,” he said. “The same is true for adding trace greenhouse gases to the atmosphere. It doesn’t take very much of an increase before their presence literally changes the color of the atmosphere as observed by infrared satellite instruments.”
Livesey says the MLS instrument has contributed to our understanding of atmospheric ozone. For example, it’s been instrumental in verifying the recovery of the ozone layer. MLS has also contributed to studies of how much stratospheric ozone descends into the lower atmosphere, contributing to surface pollution. Surface-level ozone pollution has a detrimental impact on plant growth, resulting in billions of dollars in estimated crop losses.
“NASA is mandated to study the upper atmosphere, and the word ozone appears in that mandate,” Livesay says. “It’s also in the U.S. Clean Air Act. NASA has spearheaded numerous ozone research campaigns and has contributed to many of the big-name atmospheric ozone models. We’ve done a lot of work on satellite measurements of air quality. And the A Train constellation of atmospheric research satellites, of which Aura/MLS is one component, has been a huge benefit to the atmospheric science community.” (Learn more about NASA's role in studying Earth's atmosphere.)
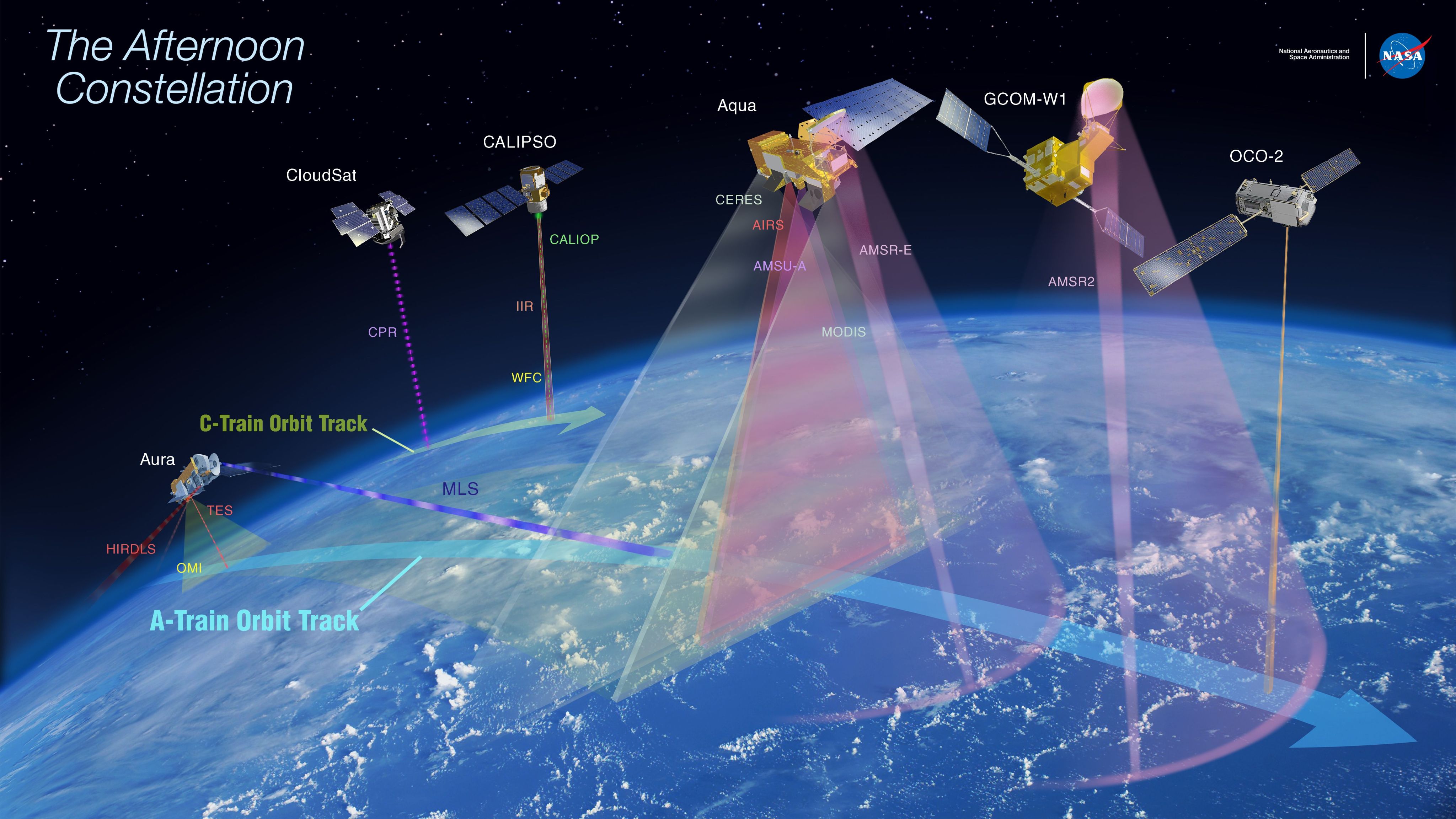
In addition to ozone, MLS tracks water vapor, numerous trace gasses and mid-atmospheric temperatures.
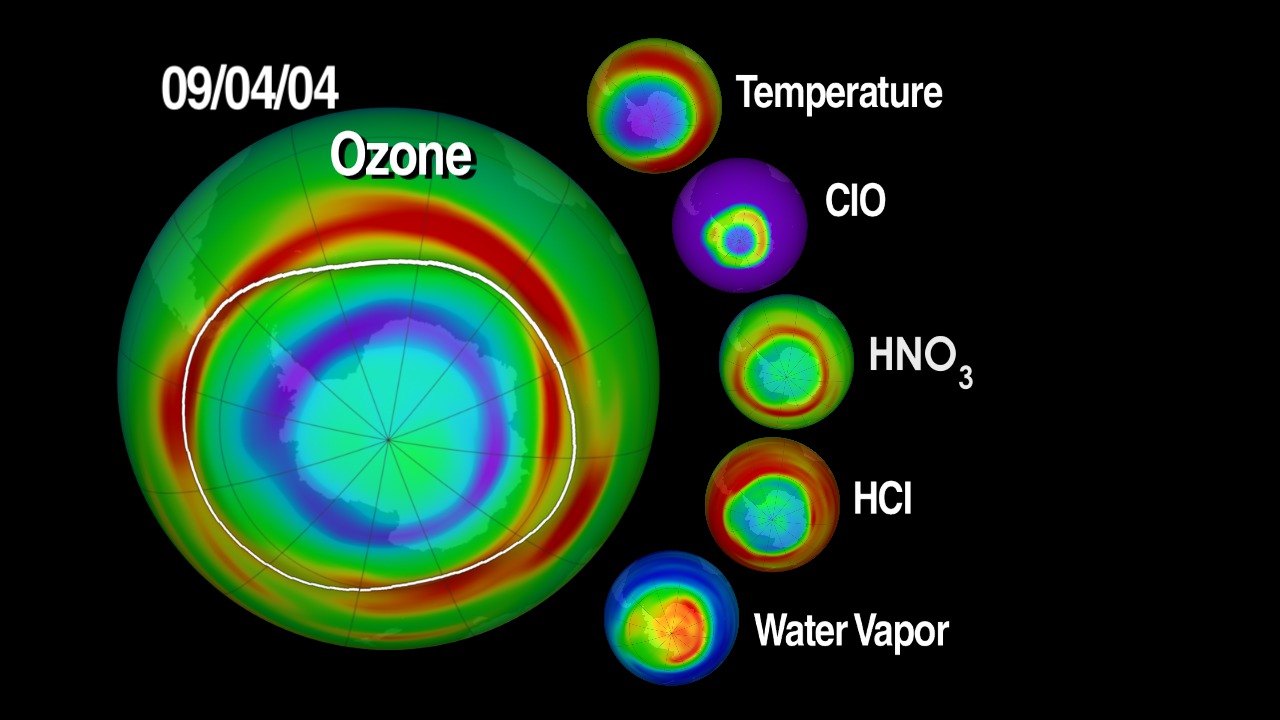
Regarding water vapor, Livesey says scientists still don’t fully understand the processes that control humidity in the stratosphere. For example, in 2000, measurements showed the amount of stratospheric water vapor decreased by about 10 percent, which slowed the rate of global surface temperature increases by about 25 percent. But scientists are still not completely sure why it decreased. “Since stratospheric water vapor is a greenhouse gas, we want to be able to predict its future evolution well,” he said. “We don’t yet fully understand the interplay of the various processes involved and how they will evolve in a warming climate. MLS data are contributing to atmospheric models that are assisting in this area of research.”
One of the biggest surprises from MLS data has been its observations of a phenomenon that allows pollution from strong forest fires to penetrate into the stratosphere. Fires are a significant contributor to stratospheric aerosols and thus have the potential to affect surface warming. “MLS has allowed us to track this pollution around the globe. We wouldn’t have guessed before that a single forest fire could do that,” Livesey said.”
As the MLS data record approaches 15 years, Livesey says he’s hopeful MLS will continue to provide important science for several more years. The instrument continues to work well and the biggest limitation on its life is the amount of fuel on the Aura spacecraft, which should run out in about 2025, although the team is considering adopting a less fuel-intensive orbit maintenance strategy that could add several more years of operations.
“The longer our data record goes, the more valuable it becomes,” he said.
For more on MLS, visit https://mls.jpl.nasa.gov/index-eos-mls.php.
Part Two of this series: 'The Atmosphere: Getting a Handle on Carbon Dioxide'